 (NHTSA) those have been almost completely eliminated, now that the public understands the need for seating children in the rear seats of vehicles, away from the airbags. However, the other injuries to drivers and front seat passengers have largely been ignored, since it is estimated that over 28,000 lives have been saved as of 2009 by using airbags.
(NHTSA) those have been almost completely eliminated, now that the public understands the need for seating children in the rear seats of vehicles, away from the airbags. However, the other injuries to drivers and front seat passengers have largely been ignored, since it is estimated that over 28,000 lives have been saved as of 2009 by using airbags.
The rapid deployment of the airbag at speeds greater than 100 mph has caused broken bones and skin abrasions. Around 2.5% of all drivers in crashes are likely to suffer serious injury as a result of airbag deployment, according to Craig Newgard, from Oregon Health & Science University, who researched statistics on 1995–2005 crashes. Burns to faces and arms, coughing fits, and asthma attacks are other common injuries during airbag deployment, according to the Automotive Occupant Restraints Council (AORC).
The hazards of airbags are not being ignored by OSHA, the Occupational Safety and Hazard Administration, the watchdog for worker’s safety. They began issuing guidelines in 2001 for emergency responders that include cautions against breathing in the contents of the passenger compartment of cars where airbags have been deployed and warning against touching the steering column due to the heat of the chemical reaction that inflates the airbag. Undeployed airbags at crash sites are also a hazard for emergency personnel if they inflate unexpectedly while the accident victim is being extracted from the car, warns OSHA.
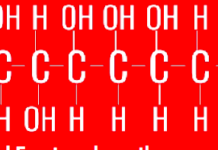
All emergency responders are now trained to deactivate undeployed airbags before treating patients in the car as advised by training groups such as Midsouth Rescue Technologies, a non-profit Fire and Rescue Training Organization.According to Washington University’s department of chemistry, sodium azide, NaN3, is the most common propellant for the airbag. When a crash occurs the azide is ignited and it decomposes to produce nitrogen gas, N2, which fills the airbag (2NaN3à 2Na + 3N2) in .025 seconds at speeds between 150 and 200 miles per hour. Extreme heat is also given off by the exothermic reaction of the sodium azide as it ignites
Sodium hydroxide, a caustic powder, is one of the by-products of the reaction. It is generated because potassium nitrate, KNO3, is placed in the detonator canister with the azide to scavenge sodium formed during the explosion. Sodium metal is extremely reactive, and can actually causing unwanted explosions with water (2Na + 2H2O –> 2NaOH + H2). The heat given off when sodium metal reacts with water is sufficient to ignite and explode the hydrogen gas produced.
Sodium hydroxide is often found in the gas fumes that escape from the airbag as it deflates. Although in only small amounts, the NHTSA has found that sodium hydroxide is causing coughing fits and asthma attacks in the drivers and passengers. Rescue workers are advised to wear gloves and safety glasses and to completely ventilate the car for several minutes before entering. The excessive coughing caused by the air bag powder can also make a chest injury worse.
Emergency personnel are now treating all accidents with airbag deployments as potential brain, chest and spine damage, according to Midsouth Rescue Technologies. Also, according to the AORC, the recommended hand position on steering wheels has changed from ten and two to nine and three o’clock in order to avoid damage to the radius and ulna, the bones in the arm and also to prevent the arm from damaging the face after it is hit by the airbag.
Others, such as University of Arizona researcher, Eric Betterton, are concerned that sodium azide will escape from the container and get into the environment. Sodium azide is an extremely toxic poison, capable of destroying entire ecosystems. An undeployed airbag contains between 50 grams for the driver side and 200 grams for the passenger side. After the airbag reacts, all of the sodium azide should be converted to harmless nitrogen gas. Undeployed airbags should be removed from cars before they are “junked” or put into landfills in order to prevent unreacted sodium azide from getting into the environment, recommends Betterton.
New types of airbags have been appearing since 1998, such as the side impact airbag and the dual depth system. Inflators using compressed nitrogen or argon gas have been replacing the sodium azide, according to Midsouth Rescue Technologies. Airbags are now being tested using crash dummies that simulate young children and smaller female passengers, instead of the previous practice of just a 5’8”, 180-pound male adult.
So how does Teresa feel about airbags? “I wouldn’t drive in a car without them. We all walked away with no broken bones while the van was totaled.”










Great description of the chemical reaction involved in this process! I had no idea how the "magic" happened! Curious whether encountering airbag deployment and the related chemicals may have long-term effects on drivers who already have asthma?
If by long time do you mean that they may have a fatal asthma attack supplimented by the panic of awaking in a smoke filled car, then yes, it can have a lasting effect. Thankfully this propellant is being phased out so the sodium hydroxide is less likely. The talc however will still be present as it lubricates the airbag and keeps it pliable. Asthmatics are sensitive to any type of particle in the air, even the chemically inert talc.
I liked the way you approached the topic and gave the specific information to back-up your article
I surely did not until I started really finding out a lot more than I ever imagined!
-Rose
Thanks for the feedback!
Thanks! I spent a lot of time researching to get my facts. I am glad that I learned how to use diigo to keep my sources straight.
Whoa, whoever knew airbags could cause danger
I think she probably had some kind of attack due to the evidence.it was great discptirion and i learnedd something new from reading this.
Teresa had an asthma attack because of the powder that is inside the airbag and comes shooting out during the deployment. Somtimes people die from asthma attacks, so they should be taken seriously. I am glad that you liked my descriptions!
I never knew how dangerous airbags can be. I thought they are suppose to protect you but i see they can also hurt you. I guess its like my mama say "I brought you into this world i can take you out." My new saying is "I can protect you, but I can also hurt you."
cool i never new how dangerous air bags were.
Many things that are designed to help us if used the wrong way can do damage to us. The deal with airbags is that they need to be investigated a little more and improved so that we can feel their full benefit.
who wouldve known all those chemicals were in things that are made to save our lives…
:wah… well thats sounds scary! i feel that they should pay more attention to the adults too because more people are being hurt nd dieing because of the air bags.. i acually learned something from this article bacuse i didnt know that an airbag could do so much damage
Yeah your right Raenishia
i knew airbags were dangerous by how they just explode out
Great intro it really makes the reader want to read more. The chemical information helped with a lot of the questions I had about air bags.
:wah:@@:cried thats horrible how do we not know dat airbaggs r dangerous thats y they have a age limit to sitting n the front seat.
Yes, don’t let any little kids or babies sit in the front seats near the airbags. Even little old ladies should probably sit in the back so their fragile bones don’t get broken.
oh my goss ! i never knew AIRBAGS can be sooo dangerous ! this gave me a heads up and a bit of a fright !:why
Just like a lot of things, there are benefits and risks to using airbags. It is important that drivers and passengers know the risks and how to prevent serious injury from airbags. Like not letting little kids sit in the front seat near the airbags.
Atleast people walk away with only broken bones. They could be seriously injured without the airbags.
I totally agree with you! Airbags have saved thousands of lives. I just think we should make them even better so that when they are saving lives they don’t harm the people they are protecting in the process.
I think thats a great idea. It’s Truly a Blessing what happened to Teresa
Yes, our entire family was glad that Teresa’s car had airbags, even though they did some damage to her.
I never knew that airbags had chemicals in them. Thats why you have to be a certain age to sit in the front. I learn something new everyday
arent ther air bags in the back of the car to
-‘ I never thought that an air bag could cause a lot of damage to peoples bodies when they are in a car accident!
Airbags are still better than not using airbags. They do protect us from a lot of damage to our bodies.
i never knew airbags were so dangerous…i thought it protected you so that u didnt hit ur face or anything so now i know the true colors of airbags…:arhh
Airbags are still better than nothing at all! It just is that they need to be made safer themselves.
good job! i never would have guessed airbags would be that dangerous!
I did not know of these dangers, as of never being in an accident myself. But i still believe they are better than being thrown outside of the car! This article gave a lot of facts, and i loved the lede. It really drew me in.
Wow i never thought airbags contained those chemicals! Very interesting and informative! Well done
great article! i never would have thought airbags were that dangerous
This is a very well-written article. I in fact knew that airbags could potentially cause harm. However, they cause more good than harm in most situations.
i agree i didnt relize how much airbags are a risk :smile:smile
Was in an accident when the airbag deployed and ripped open. Felt like my lungs were on fire. Have had serious respiratory illnesses and pneumonia muliple times since that date and left scar tissue in my lungs. Glad to still be here, but have been very sickly since the accident.
Great article never knew this. Helped us in science a lot[quote][/quote] :smile
I believe, after some hard probing, that this article is very accessible and reliable
WOW this article is amazing, never knew the things that were ment to help actually hurt us. thanks a lot
I was in 2006 Tahoe when I had so they say a head on collision with an 18 wheeler I remember nothing. I think seat belt saved me but also set me up.Airbag deployed at steering wheel broke all my ribs broke pelvis and ribs puncher ed and collapse both lungs. I’m still alive they say
:congrate :ak WOW
Approx 12 yrs ago I had a 1994 ? volvo air bag deploy and was diagnosed 1 yr. ago with lung cancer (never smoked) maybe it was one of the contributing factors???
Interesting article. I was in a mva a year ago; still have croupy, productive cough. Had to use a dishtowel to spit into (sorry yes gross) & when dried I saw glitter in the dried secretions. Very little literature out there considering how many years we have had airbags, of peoples issues afterwards. Are there recommended changes to stop using sodium azide actually that have taken place ? Or are they still using known substances and cancelling any chat about them? I am looking for answers how to be healed. I no longer get deep sleep due to this constant cough— & affects husbands sleep too. Anyone else have issues?
In 2019 I was a passenger in an automobile accident. I had 6 fractures in my skull from the front and side airbags. The skin on the tip of my nose/part of my right cheek was gone. Now that has healed, however since that I have strange fibers that are still working their way to the surface of my right cheek in 3 places where the airbag/propellant hit my face. My lungs are still affected as well. My question is… what is this that periodically works it’s way to the surface?
Is anyone still monitoring this. I have some questions about my husband who was in a vehicle accident and the airbag deployed on him he has some health issue is going on that are unexplained they cannot find what the cause of his issues are.